| Identification | Back Directory | [Name]
Peramivir | [CAS]
330600-85-6 | [Synonyms]
Bcx-1812
Rapiacta
Peramivir
PeraMiviv
Rwj-270201
Aids114230
Aids-114230
Peramivir(BCX-1812)
(229614-55-5) peramivir
PeraMivir(RWJ-270201,BCX-1812)
(1S,2S,3R,4R)-3-((S)-1-AcetaMido-2-ethylbutyl)-4-guanidino-2-hydroxycyclopentanecarboxylic acid
(1S,2R,3R,4R)-3-(1-acetaMido-2-ethyl-butyl)-4-(diaMinoMethylideneaMino)-2-hydroxy-cyclopentane-1-car
(1S,2R,3R,4R)-3-(1-acetamido-2-ethyl-butyl)-4-(diaminomethylideneamino)-2-hydroxy-cyclopentane-1-carboxylic acid
(1S,2S,3R,4R)-3-[(1S)-1-(Acetylamino)-2-ethylbutyl]-4-[(aminoiminomethyl)amino]-2-hydroxycyclopentanecarboxylic acid
Cyclopentanecarboxylic acid, 3-[(1S)-1-(acetylamino)-2-ethylbutyl]-4-[(aminoiminomethyl)amino]-2-hydroxy-, (1S,2S,3R,4R)- | [Molecular Formula]
C15H28N4O4 | [MDL Number]
MFCD09837902 | [MOL File]
330600-85-6.mol | [Molecular Weight]
328.41 |
| Chemical Properties | Back Directory | [Melting point ]
170 - 172°C (dec.) | [density ]
1.39 | [storage temp. ]
Sealed in dry,2-8°C | [solubility ]
Methanol (Slightly, Heated), Water (Slightly) | [form ]
Solid | [pka]
4.08±0.70(Predicted) | [color ]
White to Off-White |
| Hazard Information | Back Directory | [Description]
Peramivir is a neuraminidase (NA) inhibitor that was approved in Japan
in 2010 for treatment of patients with influenza. It is the only NA inhibitor
available for IV use and is the first of two NA inhibitors approved in 2010, the
second being the inhaled drug laninamivir octanoate .
Peramivir is the only NA inhibitor approved for IV use,which gives it a unique place in
influenza treatment for seriously ill patients. Peramivir was discovered
using structure-based drug design and is synthesized in six steps from
Boc-protected methyl (1S,4R)-4-amino-cyclopent-2-enecarboxylate, which
is prepared from 2-azabicyclo[2.2.1]hept-5-en-3-one. Cycloaddition
of the cyclopentene olefin with a nitrile oxide provided an intermediate
fused cyclopentane-dihydroisoxazole. Hydrogenolysis and acetylation set
up a fully functionalized cyclopentane with all four stereocenters established.
Deprotection of the amine and acid groups was followed by installation
of the guanidine moiety to provide peramivir. Like zanamivir and
oseltamivir, peramivir is a potent inhibitor of influenza virus A and B NA
[strain A(H1N1) IC50= 0.34 nM; strain A(H3N2) IC50= 0.60 nM; strain B
IC50= 1.36 nM]. However, peramivir is less potent against oseltamivirresistant
viruses that have the H275Y NA mutation. These viruses remain
sensitive to zanamivir. Peramivir is active against influenza A and B viruses
and has a lowenzymatic off-rate, suggesting that it could inhibitNAactivity
for a prolonged period and allow lower frequency of dosing. Peramivir has
proven efficacious in preclinical animal models of influenza infection. | [Chemical Properties]
White to Off-White Solid | [Originator]
BioCryst Pharmaceuticals Inc. (United States) | [Uses]
Peramivir is a new antiviral agent for influenza treatment; it can be used as neuraminidase inhibitor for treating human and avian influenza
| [Definition]
Peramivir is a member of the class of guanidines that is used (as its trihydrate) for the treatment of acute uncomplicated influenza in patients 18 years and older who have been symptomatic for no more than two days.
| [Brand name]
Rapiacta, PeramiFlu | [Synthesis]
Several syntheses of this drug have been reported and the improved route disclosed in a recent
patent is described in the scheme. Ring opening of commercially available (?à)-2-
azabicyclo[2.2.1]hept-5-en-3-one (117) with methanolic HCl followed by classical resolution with Ltartaric
acid gave the (1S,4R)-methyl ester 118 in 85% yield. Protection of 118 with Boc anhydride and
TEA in CH2Cl2 afforded carbamate 119 in 90% yield. Alkene 119 was then subject to nitrone dipolar
cycloaddition conditions involving 2-ethyl-N-hydroxybutanimidoyl chloride 120 and triethylamine,
followed by the basic workup and then treatment with methanolic HCl, ultimately resulting in
dihydroisoxazole 121. Interestingly, the nitrone generated from 120 approached alkene 119 from the
less hindered face and proceeded with remarkable regioselectivity to provide azacycle 121 in 76% yield
for the three step sequence. Treatment of 121 with 1.5 eq. lithium aluminum hydride resulted in
rupture of the N-O bond within this system, which afforded the amino alcohol 122 in 81% yield. It
should be noted that neither the Boc group or the methyl ester were reduced under these reaction
conditions. Then, a one-pot three step sequence involving acetylation of the amino group, removal of
the Boc group, and hydrolysis of the carboxylic ester followed by guanylation with
pyrazolecarboxamidine hydrochloride (123) provided peramivir (X) in 82% yield over the final four
steps.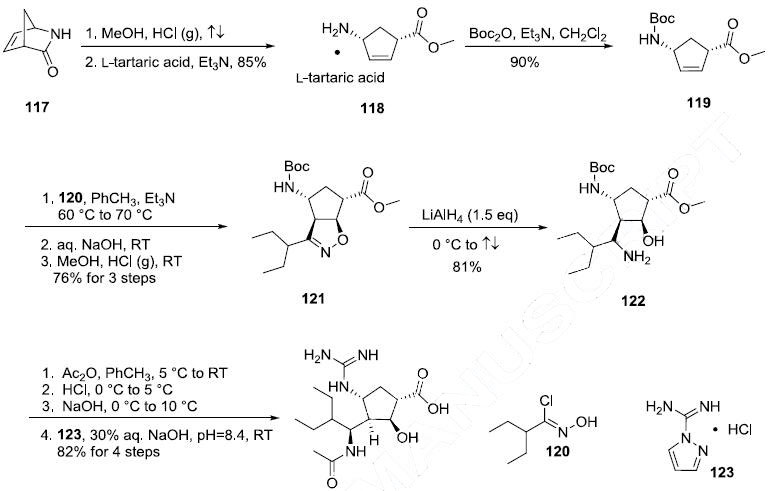
|
|
|