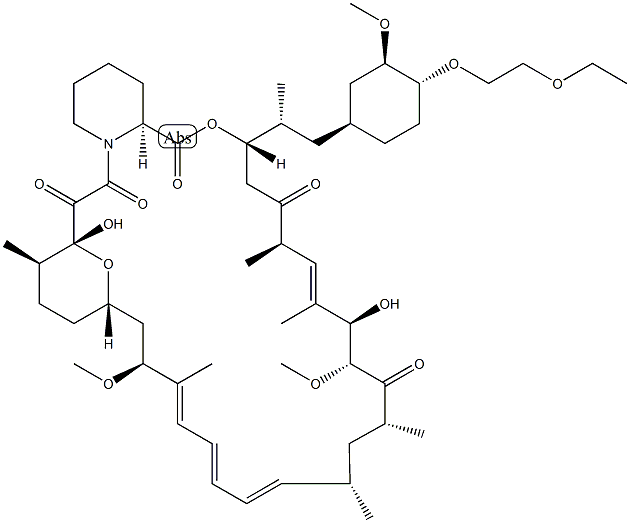
UMiroliMus
- CAS No.
- 851536-75-9
- Chemical Name:
- UMiroliMus
- Synonyms
- TRM986;TRM 986;TRM-986;BioliMus;UMiroliMus;BIOLIMUS A9;BioliMus A9 UMiroliMus;BioliMus, BioliMus A9, TRM 986;Rapamycin, 42-O-(2-ethoxyethyl)-
- CBNumber:
- CB22470849
- Molecular Formula:
- C55H87NO14
- Molecular Weight:
- 986.28
- MDL Number:
- MFCD22124433
- MOL File:
- 851536-75-9.mol
| Boiling point | 991.0±75.0 °C(Predicted) |
|---|---|
| Density | 1.15±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMF: Soluble; DMSO: Soluble; Ethanol: Soluble; Methanol: Soluble |
| form | A solid |
| pka | 10.40±0.70(Predicted) |
| FDA UNII | U36PGF65JH |
UMiroliMus price More Price(6)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Cayman Chemical | 23585 | Umirolimus ≥95% | 851536-75-9 | 1mg | $117 | 2024-03-01 | Buy |
| Cayman Chemical | 23585 | Umirolimus ≥95% | 851536-75-9 | 5mg | $463 | 2024-03-01 | Buy |
| TRC | U710005 | Umirolimus | 851536-75-9 | 5mg | $370 | 2021-12-16 | Buy |
| TRC | U710005 | Umirolimus | 851536-75-9 | 1mg | $85 | 2021-12-16 | Buy |
| American Custom Chemicals Corporation | API0007715 | UMIROLIMUS 95.00% | 851536-75-9 | 5MG | $504 | 2021-12-16 | Buy |
UMiroliMus Chemical Properties,Uses,Production
Description
Coronary stents have dramatically improved the success rate of interventional cardiology in recent years, and stent implantation has become the standard of care in percutaneous coronary interventions. However, the long-term success of coronary stenting is hampered by a high rate of restenosis (i.e., recurrence of stenosis, or reblocking), which is caused by proliferation and migration of smooth muscle cells and production of extracellular matrix. The Biomatrix DES is a novel stent system combining a biodegradable PLA and the new anti-restenoic drug biolimus. Biolimus is a semi-synthetic analog of sirolimus wherein the hydroxyl moiety at position 42 is modified to an ethoxyethyl ether group. As with rapamycin, the mechanism of action of biolimus consists of forming a complex with intracellular 12-kDa FK506-binding protein (FKBP-12), which binds to the mammalian target of rapamycin (mTOR) and reversibly inhibits cell-cycle transition of proliferating smooth muscle cells.The antiproliferative potency of biolimus is similar to that of sirolimus; however, it is approximately 10-fold more lipophilic than sirolimus, which results in rapid absorption of the drug into fatty tissues and reduced systemic exposure. The Biomatrixs DES is produced by the absorption of a 1:1 combination of biolimus and PLA on a flexible stainless steel stent. The precision automated coating method used in the production of the stent ensures the PLA biolimus combination is applied solely to the abluminal surface of the stent. PLA is co-released with biolimus over 6 9 months, and biodegraded initially to lactic acid, and eventually to carbon dioxide and water.
Originator
Biosensors (United States)
Uses
Umirolimus is a semi-synthetic macrocyclic lactone prepared from rapamycin by selective alkylation of the 42-hydroxy group, providing one of most hydrophobic tacrolimus analogues. Umirolimus has been targeted for use in stents and medical devices to suppress localised immunoreaction. Like all tacrolimus analogues, umirolimus binds to receptor protein, FKBP12. The complex then binds to mTOR and prevents it from interacting with target proteins. Umirolimus is extensively cited in the literature with over 70 citations.
Uses
Umirolimus is one of two promising agents for the treatment of peripheral artery disease (PAD) and minimize restenosis. Umirolimus is a medical device to suppress localised immunoreaction.
brand name
Biomatrix
Synthesis
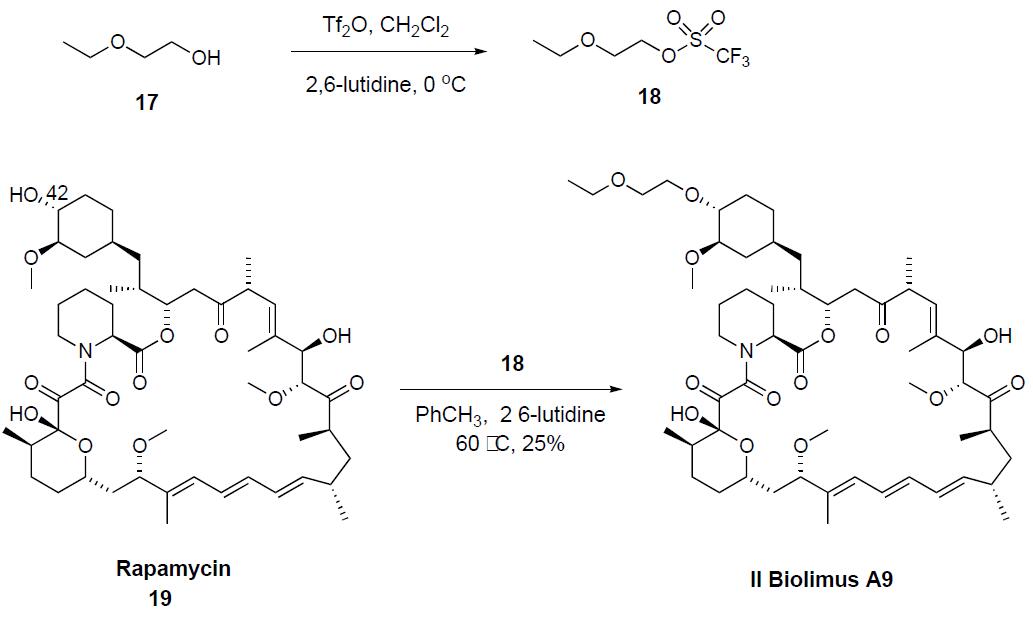
The synthesis involves the preparation of the ethoxyethyl triflate 18 which was obtained by reacting ethoxyethanol 17 with triflic anhydride in dichloromethane in the presence of 2,6-lutidine at 0 ??C. Selective alkylation of rapamycin 19 alcohol at C-42 with crude triflate 18 in a mixture of toluene and 2,6-lutidine at 60 ??C gave biolimus A9 II in 25% isolated yield.
UMiroliMus Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 | marketing@targetmol.com | United States | 19892 | 58 |
| BOC Sciences | +16314854226 | inquiry@bocsci.com | United States | 19743 | 58 |
| Aladdin Scientific | +1-833-552-7181 | sales@aladdinsci.com | United States | 52927 | 58 |
| LGM Pharma | 1-(800)-881-8210 | inquiries@lgmpharma.com | United States | 2127 | 70 |
| ChemStrong Scientific Co.,Ltd | 0755-0755-66853366 13670046396 | sales@chem-strong.com | China | 17994 | 56 |
| Lynnchem | 86-(0)29-85992781 17792393971 | info@lynnchem.com | China | 4587 | 58 |
| Novachemistry | 44-20819178-90 02081917890 | info@novachemistry.com | United Kingdom | 4381 | 58 |
| Wuhan FengyaoTonghui Chemical Products Co., Ltd. | 027-87466105 15377573527 | 2678564200@qq.com | China | 17997 | 58 |
| Shanghai Hongye Biotechnology Co. Ltd | 400-9205774 | sales@glpbio.cn | China | 6870 | 58 |
| Supplier | Advantage |
|---|---|
| Hubei Jusheng Technology Co.,Ltd. | 58 |
| TargetMol Chemicals Inc. | 58 |
| BOC Sciences | 58 |
| Aladdin Scientific | 58 |
| LGM Pharma | 70 |
| ChemStrong Scientific Co.,Ltd | 56 |
| Lynnchem | 58 |
| Novachemistry | 58 |
| Wuhan FengyaoTonghui Chemical Products Co., Ltd. | 58 |
| Shanghai Hongye Biotechnology Co. Ltd | 58 |