1,5-ジイソシアナトナフタレン 化学特性,用途語,生産方法
外観
白色~うすい黄色粉末~塊
用途
塗料?接着剤?ウレタンエラストマー原料
説明
Naphthalene diisocyanate (NDI) occurs as white to lightyellow
crystalline flakes with a characteristic odor. NDI is
incompatible with many classes of compounds, reacting
exothermically to release toxic gases. Reactions with amines,
aldehydes, alcohols, alkali metals, ketones, mercaptans, strong
oxidizers, hydrides, phenols, and peroxides can cause vigorous
releases of heat. Acids and bases initiate polymerization reactions.
NDI can react with water to form amines and liberate
carbon dioxide.
化学的特性
Naphthylene 1,5-diisocyanate is a solid, m.p. 128°C. It has a lower vapour
pressure than tolylene diisocyanate and is therefore less toxic in use; it does,
however, have sensitizing properties.
使用
Manufacture of polyurethane solid elastomers.
製造方法
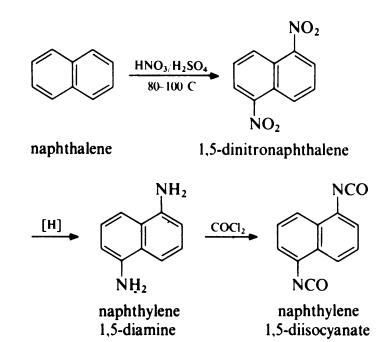
Naphthylene 1,5-diisocyanate (NDI) is prepared from naphthalene as follows:
一般的な説明
White to light-yellow crystalline flakes.
反応プロフィール
Isocyanates and thioisocyanates, such as 1,5-Naphthalene diisocyanate, are incompatible with many classes of compounds, reacting exothermically to release toxic gases. Reactions with amines, aldehydes, alcohols, alkali metals, ketones, mercaptans, strong oxidizers, hydrides, phenols, and peroxides can cause vigorous releases of heat. Acids and bases initiate polymerization reactions in these materials. Some isocyanates react with water to form amines and liberate carbon dioxide. Base-catalysed reactions of isocyanates with alcohols should be carried out in inert solvents. Such reactions in the absence of solvents often occur with explosive violence, [Wischmeyer(1969)].
危険性
Irritant. Questionable carcinogen.
安全性プロファイル
A powerful allergen. An
irritant. Questionable carcinogen. When
heated to decomposition it emits toxic
fumes of NOx.
環境運命予測
NDI is a synthetic organic chemical. It is a natural derivative of
primary amines with the general formula R–N]C]O which
does not occur naturally in the environment. At room temperature
it can be a liquid or crystal. It is miscible with alcohol,
diglycol, monoethyl ether, ether, acetone, carbon tetrachloride,
benzene, chlorobenzene, kerosene, and olive oil; however, it
may react violently with alcohol, water, acid, bases, and strong
alkaline materials and tertiary amines and generate enough heat
to self-ignite and release toxic combustion products. NDI is not
readily biodegradable; however, it reacts with water and most
acids producing unstable carbonic acids, which subsequently
decarboxylate yielding relatively chemically inert and insoluble
polymeric urea. While these polyureas are persistent, studies
have indicated that they pose virtually no potential for adverse
impacts on the aquatic environment. Due to hydrolysis in water,
bioaccumulation of NDI is not expected. Since the hydrolysis
products formed are irritants, there is a potential for inhalation
exposure. The degree stability is a function of humidity.
1,5-ジイソシアナトナフタレン 上流と下流の製品情報
原材料
準備製品