|
|
| | SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE Basic information |
| Product Name: | SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE | | Synonyms: | Phosphoricacid,disodiumsalt,heptahydrate;Sodiummonohydrogenphosphateheptahydrate(2:1:1:7);MONOHYDROGEN SODIUM PHOSPHATE;SEC-SODIUM PHOSPHATE HEPTAHYDRATE;SECONDARY SODIUM PHOSPHATE;SECONDARY SODIUM PHOSPHATE HEPTAHYDRATE;SODIUM MONOHYDROGEN PHOSPHATE HEPTAHYDRATE;SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE | | CAS: | 7782-85-6 | | MF: | H6NaO5P | | MW: | 140.01 | | EINECS: | 616-512-8 | | Product Categories: | | | Mol File: | 7782-85-6.mol | 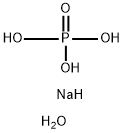 |
| | SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE Chemical Properties |
| Melting point | 48 °C | | density | 1.68 g/mL at 25 °C(lit.) | | vapor density | 4.9 (vs air) | | storage temp. | Store at +5°C to +30°C. | | solubility | 154g/l | | form | Solid | | color | White | | Specific Gravity | 1.7 | | PH Range | 8.7 - 9.3 | | PH | 8.7-9.3 (25℃, 5% in solution) | | Water Solubility | Soluble in water and insoluble in ethanol. | | Merck | 14,8659 | | LogP | -2.148 (est) | | CAS DataBase Reference | 7782-85-6(CAS DataBase Reference) | | EPA Substance Registry System | Disodium phosphate heptahydrate (7782-85-6) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36-24/25 | | RIDADR | UN 3077 9 / PGIII | | WGK Germany | 2 | | RTECS | WC4600000 | | TSCA | Yes | | HS Code | 28352990 | | Toxicity | LD50 orally in Rabbit: 12930 mg/kg |
| | SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE Usage And Synthesis |
| Chemical Properties | The USP 32 states that dibasic sodium phosphate is dried or
contains, 1, 2, 7, or 12 molecules of water of hydration.
Anhydrous dibasic sodium phosphate occurs as a white powder.
The dihydrate occurs as white or almost white, odorless crystals.
The heptahydrate occurs as colorless crystals or as a white granular
or caked salt that effloresces in warm, dry air. The dodecahydrate
occurs as strongly efflorescent, colorless or transparent crystals. | | Uses | Sodium Phosphate Dibasic Heptahydrate is used as precursor in the preparation of cobalt-based molecular catalyst for water oxidation. | | Uses | Buffering agent. | | Uses | Sodium phosphate dibasic heptahydrate has been used to prepare sodium phosphate buffer and phosphate-buffered saline (PBS) buffer.[44} | | Production Methods | Either bone phosphate (bone ash), obtained by heating bones to
whiteness, or the mineral phosphorite is used as a source of tribasic
calcium phosphate, which is the starting material in the industrial
production of dibasic sodium phosphate.
Tribasic calcium phosphate is finely ground and digested with
sulfuric acid. This mixture is then leached with hot water and
neutralized with sodium carbonate, and dibasic sodium phosphate
is crystallized from the filtrate. | | Pharmaceutical Applications | Dibasic sodium phosphate is used in a wide variety of pharmaceutical
formulations as a buffering agent and as a sequestering agent.
Therapeutically, dibasic sodium phosphate is used as a mild laxative
and in the treatment of hypophosphatemia.
Dibasic sodium phosphate is also used in food products; for
example as an emulsifier in processed cheese. | | Safety | Dibasic sodium phosphate is widely used as an excipient in
parenteral, oral, and topical pharmaceutical formulations.
Phosphate occurs extensively in the body and is involved in
many physiological processes since it is the principal anion of
intracellular fluid. Most foods contain adequate amounts of
phosphate, making hypophosphatemia (phosphate deficiency)
virtually unknown except for certain disease states or in patients
receiving total parenteral nutrition. Treatment is usually by the oral
administration of up to 100 mmol of phosphate daily.
Approximately two-thirds of ingested phosphate is absorbed
from the gastrointestinal tract, virtually all of it being excreted in the
urine, and the remainder is excreted in the feces.
Excessive administration of phosphate, particularly intravenously,
rectally, or in patients with renal failure, can cause
hyperphosphatemia that may lead to hypocalcemia or other severe
electrolyte imbalances. Adverse effects occur less frequently
following oral consumption, although phosphates act as mild saline
laxatives when administered orally or rectally. Consequently,
gastrointestinal disturbances including diarrhea, nausea, and
vomiting may occur following the use of dibasic sodium phosphate
as an excipient in oral formulations. However, the level of dibasic
sodium phosphate used as an excipient in a pharmaceutical
formulation is not usually associated with adverse effects.
LD50 (rat, oral): 17 g/kg | | storage | The anhydrous form of dibasic sodium phosphate is hygroscopic.
When heated to 40℃, the dodecahydrate fuses; at 100℃ it loses its
water of crystallization; and at a dull-red heat (about 240℃) it is
converted into the pyrophosphate, Na4P2O7. Aqueous solutions of
dibasic sodium phosphate are stable and may be sterilized by
autoclaving.
The bulk material should be stored in an airtight container, in a
cool, dry place. | | Incompatibilities | Dibasic sodium phosphate is incompatible with alkaloids, antipyrine,
chloral hydrate, lead acetate, pyrogallol, resorcinol and
calcium gluconate, and ciprofloxacin. Interaction between calcium and phosphate, leading to the formation of insoluble
calcium-phosphate precipitates, is possible in parenteral admixtures. | | Regulatory Status | GRAS listed. Accepted in Europe for use as a food additive.
Included in the FDA Inactive Ingredients Database (injections;
infusions; nasal, ophthalmic, oral, otic, topical, and vaginal
preparations). Included in nonparenteral and parenteral medicines
licensed in the UK. Included in the Canadian List of Acceptable
Non-medicinal Ingredients. |
| | SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE Preparation Products And Raw materials |
|