- Dibutylurea
-

- $8.80 / 1KG
-
2019-07-06
- CAS: 1792-17-2
- Min. Order: 1KG
- Purity: 97%-99%
- Supply Ability: 100kg
|
| | Dibutylurea Basic information |
| Product Name: | Dibutylurea | | Synonyms: | 1,3-DIBUTYLUREA;n,n’-dibutylurea(asimpurity);Urea, N,N'-dibutyl-;urea,1,3-dibutyl-;N,N'-DI-N-BUTYLUREA;N,N'-DIBUTYLUREA;TIMTEC-BB SBB007953;Dibutylurea,98% | | CAS: | 1792-17-2 | | MF: | C9H20N2O | | MW: | 172.27 | | EINECS: | 217-258-7 | | Product Categories: | Agro-Products, Aliphatics, Metabolites & Impurities, Pharmaceuticals, Intermediates & Fine Chemicals | | Mol File: | 1792-17-2.mol | 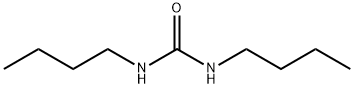 |
| | Dibutylurea Chemical Properties |
| Melting point | 65-75 °C | | Boiling point | 115°C 0,03mm | | density | 0.9845 (rough estimate) | | refractive index | 1.5800 (estimate) | | Fp | 115°C/0.03mm | | storage temp. | Sealed in dry,Room Temperature | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | pka | 14.44±0.46(Predicted) | | color | White to Off-White | | BRN | 1704165 | | CAS DataBase Reference | 1792-17-2(CAS DataBase Reference) | | NIST Chemistry Reference | 1,3-Dibutylurea(1792-17-2) | | EPA Substance Registry System | N,N'-Dibutylurea (1792-17-2) |
| | Dibutylurea Usage And Synthesis |
| Chemical Properties | white to pink or light beige crystalline powder | | Uses | N,N''-Dibutylurea is a degradation product of Benomyl, the active ingredient in Benlate fungicides. | | Uses | N,N'-Dibutylurea is a degradation product of Benomyl, the active ingredient in Benlate fungicides. | | Preparation | The reactions under CO2 pressured conditions were carried out in a stainless steel reactor (SUS 304, 30 mL, TVS-5 type). Thus, butylamine (2.9 g, 40 mmol), Ph3SbO (370 mg, 1.0 mmol), P4S10 (890 mg, 2.0 mmol), and benzene (20 mL) were charged into the reactor, and then CO2 was introduced at a pressure of 4.9 MPa (50 kg cm-2, ca. 65 mmol) at room temperature. The reactor was heated at 80 ℃ in a temperature-regulated incubator for 12 h. When a reaction temperature higher than 100 ℃ was necessary, an oil bath was used. After the heating, the reactor was cooled and carefully decompressed. The contents were treated with hot benzene (3×20 mL) and filtered to remove an insoluble residue containing the catalyst and phosphoric acid derivatives. The collected benzene solution was then concentrated to dryness in vacuo with cooling. Pure 1,3-dibutylurea was isolated by recrystallization from ligroin (yield 2.99 g, 88%). | | Synthesis Reference(s) | Synthetic Communications, 21, p. 1923, 1991 DOI: 10.1080/00397919108021783 |
| | Dibutylurea Preparation Products And Raw materials |
| Raw materials | Ethanediamide,N1,N2-dibutyl--->1-benzyl-3-butyl-1-methyl-urea-->4-(BUTYLCARBAMOYL)MORPHOLINE-->N-butyl-N'-phenylurea-->Urea, N,N,N'-tributyl--->BIS(2-(METHOXYCARBONYL)PHENYL) CARBONATE-->Butylamine Hydrobromide-->Butyl isocyanate-->N-Methylbenzylamine-->Carbon tetrabromide-->Dibutylamine-->Aniline-->carbon monoxide | | Preparation Products | 1,3,5-Triazine-2,4,6(1H,3H,5H)-trione, 1,3-dibutyl- |
|