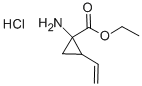
Cyclopropanecarboxylic acid, 1-amino-2-ethenyl-, ethyl ester, hydrochloride (9CI) synthesis
- Product Name:Cyclopropanecarboxylic acid, 1-amino-2-ethenyl-, ethyl ester, hydrochloride (9CI)
- CAS Number:681807-60-3
- Molecular formula:C8H13NO2.HCl
- Molecular Weight:0
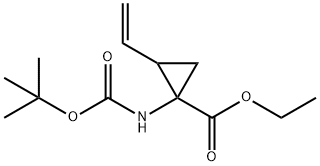
681807-59-0
72 suppliers
inquiry

681807-60-3
16 suppliers
inquiry
Yield:681807-60-3 100%
Reaction Conditions:
with hydrogenchloride in 1,4-dioxane at 20; for 2 h;
Steps:
B.2.A2
Example 2 The desired product was made by each of the following methods: Method A Glycine ethyl ester hydrochloride (303.8 g, 2.16 mol) was suspended in tert-butylmethyl ether (1.6 L). Benzaldehyde (231 g, 2.16 mol) and anhydrous sodium sulfate (154.6 g, 1.09 mol) were added and the mixture cooled to 0° C. using an ice-water bath. Triethylamine (455 mL, 3.26 mol) was added dropwise over 30 minutes and the mixture stirred for 48 hours at room temperature. The reaction was then quenched by addition of ice-cold water (1 L) and the organic layer was separated. The aqueous phase was extracted with tert-butylmethyl ether (0.5 L) and the combined organic phases washed with a mixture of saturated aqueous NaHCO3 (1 L) and brine (1 L). The solution was dried over MgSO4, filtered, and concentrated in vacuo to afford 392.4 g of the desired product as a thick yellow oil that was used directly in the next step. 1H NMR (CDCl3, 300 MHz) δ 1.32 (t, J=7.1 Hz, 3H), 4.24 (q, J=7.1 Hz, 2H), 4.41 (d, J=1.1 Hz, 2H), 7.39-7.47 (m, 3H), 7.78-7.81 (m, 2H), 8.31 (s, 1H). To a suspension of lithium tert-butoxide (84.06 g, 1.05 mol) in dry toluene (1.2 L), was added dropwise a mixture of the product of Step A1 (100.4 g, 0.526 mol) and trans-1,4-dibromo-2-butene (107.0 g, 0.500 mol) in dry toluene (0.6 L) over 60 minutes. After completion of the addition, the deep red mixture was quenched by addition of water (1 L) and tert-butylmethyl ether (TBME, 1 L). The aqueous phase was separated and extracted a second time with TBME (1 L). The organic phases were combined, 1N HCl (1 L) was added, and the mixture stirred at room temperature for 2 hours. The organic phase was separated and extracted with water (0.8 L). The aqueous phases were then combined, saturated with salt (700 g), TBME (1 L) was added and the mixture cooled to 0° C. The stirred mixture was then basified to pH 14 by the dropwise addition of 10N NaOH, the organic layer separated, and the aqueous phase extracted with TBME (2×500 mL). The combined organic extracts were dried (MgSO4), filtered, and concentrated to a volume of 1 L. To this solution was added di-tert-butyldicarbonate (131.0 g, 0.6 mol) and the mixture stirred 4 days at room temperature. Additional di-tert-butyldicarbonate (50 g, 0.23 mol) was added to the reaction, the mixture refluxed for 3 hours, and was then allowed cool to room temperature overnight. The reaction mixture was dried over MgSO4, filtered, and concentrated in vacuo to afford 80 g of crude material. This residue was purified by flash chromatography (2.5 Kg of SiO2, eluted with 1% to 2% CH3OH/CH2Cl2) to afford 57 g (53%) of racemic product as a yellow oil which solidified while sitting in the refrigerator. 1H NMR (CDCl3, 300 MHz) δ 1.26 (t, J=7.1 Hz, 3H), 1.46 (s, 9H), 1.43-1.49 (m, 1H), 1.76-1.82 (br m, 1H), 2.14 (q, J=8.6 Hz, 1H), 4.18 (q, J=7.2 Hz, 2H), 5.12 (dd, J=10.3, 1.7 Hz, 1H), 5.25 (br s, 1H), 5.29 (dd, J=17.6, 1.7 Hz, 1H), 5.77 (ddd, J=17.6, 10.3, 8.9 Hz, 1H); MS m/z 254.16 (M+-1). The product of Step A2 (9.39 g, 36.8 mmol) was dissolved in 4N HCl/dioxane (90 mL, 360 mmol) and was stirred for 2 hours at room temperature. The reaction mixture was concentrated to supply the desired product in quantitative yield (7 g, 100%). 1H NMR (CD3OD) δ 1.32 (t, J=7.1, 3H), 1.72 (dd, J=10.2, 6.6 Hz, 1H), 1.81 (dd, J=8.3, 6.6 Hz, 1H), 2.38 (q, J=8.3 Hz, 1H), 4.26-4.34 (m, 2H), 5.24 (dd, 10.3, 1.3 Hz, 1H) 5.40 (d, J=17.2, 1H), 5.69-5.81 (m, 1H).
References:
US2006/183694,2006,A1 Location in patent:Page/Page column 35-36
![Cyclopropanecarboxylic acid, 2-ethenyl-1-[(phenylmethylene)amino]-, ethyl ester](/CAS/20210305/GIF/1269195-07-4.gif)
1269195-07-4
0 suppliers
inquiry

681807-60-3
16 suppliers
inquiry
![Cyclopropanecarboxylic acid, 1-[(diphenylmethylene)amino]-2-ethenyl-, ethyl ester](/CAS/20210305/GIF/1023795-84-7.gif)
1023795-84-7
0 suppliers
inquiry

681807-60-3
16 suppliers
inquiry

787548-29-2
35 suppliers
inquiry

681807-60-3
16 suppliers
inquiry