| Identification | More | [Name]
Diphenhydramine | [CAS]
58-73-1 | [Synonyms]
AURORA KA-7664
DIPHENHYDRAMINE
DIPHENHYDRAMINE BASE
n,n-dimethyl-4,4-diphenyl-3-oxabutylamine
2-(benzhydryloxy),-n,n-dimethylethylamine
2-(Benzhydryloxy)-N,N-dimethylethanamine
2-(Benzhydryloxy)-N,N-Dimethylethylamine
2-(Benzohydryloxy)-N,N-dimethylethylamine
2-(diphenylmethoxy)-n,n-dimethyl-ethanamin
2-(Diphenylmethoxy)-N,N-dimethylethanam-ine
2-(diphenylmethoxy)-n,n-dimethyl-ethylamin
2-(Diphenylmethoxy)-N,N-dimethylethylamine
2-(Diphenylmethoxy)N,N-dimethylethylamine
2(diphenylmethoxy)-n,n-dimethylethylamine
Aleryl
Alledryl
Allergan B
Allergeval
Allergical
Allergival | [EINECS(EC#)]
200-396-7 | [Molecular Formula]
C17H21NO | [MDL Number]
MFCD00274173 | [Molecular Weight]
255.35 | [MOL File]
58-73-1.mol |
| Chemical Properties | Back Directory | [Melting point ]
167-172°C | [Boiling point ]
bp2.0 150-165° | [density ]
0.9889 (rough estimate) | [refractive index ]
1.5450 to 1.5490 | [storage temp. ]
Keep in dark place,Inert atmosphere,Room temperature | [solubility ]
Chloroform (Slightly), Methanol (Slightly) | [form ]
Oil | [pka]
pKa 9.1 (Uncertain) | [color ]
Oil | [Merck ]
14,3309 | [BCS Class]
1 | [Contact allergens]
This antihistaminic drug with sedative properties is
mainly sold over the counter. It can be used both topically
(treatment of pruritis) and orally for its antiallergic,
antiemetic, sedative, and anticough properties.
Allergic or photoallergic contact dermatitis and fixeddrug
eruption seem to be rare. | [CAS DataBase Reference]
58-73-1(CAS DataBase Reference) | [NIST Chemistry Reference]
Ethylamine, 2-diphenylmethoxy-n,n-dimethyl-(58-73-1) | [EPA Substance Registry System]
Ethanamine, 2-(diphenylmethoxy)-N,N-dimethyl- (58-73-1) |
| Safety Data | Back Directory | [RTECS ]
KR6825000 | [HS Code ]
2922.19.7000 | [Safety Profile]
Deadly human poison
by an unspecified route. Poison by
ingestion, intravenous, intraperitoneal, and
subcutaneous routes. Experimental
reproductive effects. Human systemic
effects by ingestion: somnolence, alteration
of operant conditioning, changes in
psychophysiological tests. Human mutation
data reported. When heated to
decomposition it emits toxic fumes of NO,.
See also ETHERS. | [Hazardous Substances Data]
58-73-1(Hazardous Substances Data) | [Toxicity]
LD50 oral in rat: 390mg/kg |
| Hazard Information | Back Directory | [Description]
Diphenhydramine is one of the main representatives of antihistamine drugs that block H1
receptors. Besides antihistamine activity, diphenhydramine exhibits a local anesthetic
effect, relaxes smooth muscle, and has sedative and soporific action. | [Originator]
Benadryl,Parke Davis,US,1946 | [Uses]
Antihistaminic. | [Uses]
Diphenhydramine also reduces muscle rigidity and general stiffness, and has a relatively
minor effect on tremors. | [Uses]
Diphenhydramine is used for symptoms of allergies, for treating hives, hay fever, serum
sickness, and other allergic illnesses, and also as a sedative and soporific drug as an independent
as well as in combination with other drugs. Synonyms of this drug are dimedrol,
benadryl, allergina, valdren, and many others. | [Definition]
ChEBI: Diphenhydramine is an ether that is the benzhydryl ether of 2-(dimethylamino)ethanol. It is a H1-receptor antagonist used as a antipruritic and antitussive drug. It has a role as a H1-receptor antagonist, an antiemetic, a sedative, an anti-allergic agent, a muscarinic antagonist, an antiparkinson drug, an antipruritic drug, a local anaesthetic, an antidyskinesia agent, an antitussive and a oneirogen. It is an ether and a tertiary amino compound. | [Manufacturing Process]
As described in US Patent 2,421,714: (a) benzhydryl
omide is first prepared
as follows: 840 parts by weight of diphenylmethane is heated to 130°C with
stirring. In the presence of a 200 watt electric light 6 inches from the flask,
880 parts of
omine is added slowly. Liberation of H
occurs and addition
requires 1 hour and 45 minutes. The temperature is maintained at 130°C for
an additional 30 minutes. A fine stream of air is blown in to remove H
and
2 while the reaction mixture cools. Benzene (180 parts) is added and the
solution used immediately in (b) below.
If pure benzhydryl
omide is desired the above reaction mixture is dissolved
in ether, washed with water, sodium carbonate solution and finally with water.
The ether is removed, benzene added and distilled off and the benzhydryl
omide distilled in vacuo. Yield 85%.
(b) 490 parts β-dimethylaminoethanol and 530 parts of anhydrous sodium
carbonate are heated to 110°C with stirring. The addition of the benzene�benzhydryl
omide mixture is then begun. The temperature is raised to 120°-
125°C. As reaction takes place carbon dioxide is evolved, the addition requires1? hours. The mixture is kept at 125°C for 5 hours additional time. After
cooling, 3,000 parts of water is added and the mixture stirred until the
inorganic salts are dissolved. The mixture is transferred to a large separatory
funnel and 1,500 parts of ether added. The ether solution is washed several
times with water and then the ether layer extracted with 1 to 4 hydrochloric
acid. The acid solution is treated with 30 parts of Darco and 30 parts Filter-Cel
and filtered.
The free base is liberated from the acid solution with 20% sodium hydroxide
solution and taken up in ether. The ether layer is washed with water, saturated
with NaCl and then shaken with solid potassium hydroxide. The ether is
removed by distillation, 200 parts of benzene added and distilled off. The
residue is distilled in vacuo and the fraction 150°-165°C/2 mm is collected
and amounts to 433 parts. The hydrochloride salt is prepared by dissolving
the free base in anhydrous ether and slowly adding an alcoholic solution of
hydrogen chloride. The solid is recrystallized from absolute alcohol-ether
mixture or isopropanol-ether mixture and has a MP of 161-162°C. | [Therapeutic Function]
Antihistaminic | [Synthesis]
Diphenhydramine, 2-diphenylmethoxy-N,N-dimethylamine (10.2.5), is
synthesized by the esterification of 2-dimethylaminoethanol with benzhydrylbromide [35¨C37].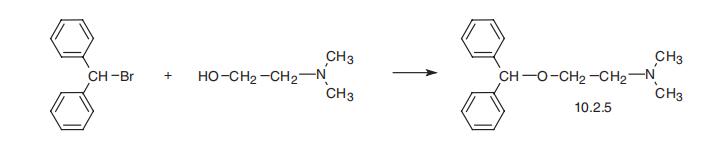
| [Environmental Fate]
Diphenhydramine is fairly stable in the environment
although it does undergo photodegradation. Conjugates of
diphenhydramine such as diphenhydramine-N-glucuronide
may be converted back to the parent compound, diphenhydramine,
through enzymatic cleavage during sewage treatment
process. Diphenhydramine is removed poorly through wastewater
treatment processes and is found in significant concentrations
in aquatic organisms downstream from such plants.
Diphenhydramine has significant risk for bioaccumulation,
particularly in water downstream from wastewater and sewage
treatment facilities. | [Toxicity evaluation]
The toxicity of antihistamines is related to their anticholinergic
(antimuscarinic), antihistamine, and serotonergic activation.
The action of acetylcholine at the muscarinic receptors is
blocked, resulting in signs and symptoms of anticholinergic
poisoning. Diphenhydramine may produce direct toxicity
unrelated to its anticholinergic properties including inhibition
of cardiac fast sodium channels and at higher concentrations,
the drug may inhibit potassium channels, which can result in
QT prolongation. Diphenhydramine also blocks the reuptake
of serotonin and has been reported to cause serotonin
syndrome in some individuals during overdose. The action of
diphenhydramine at H-1 receptors causes sedation. |
|
|