ヘリウム 化学特性,用途語,生産方法
性質
ヘリウムは、希ガス元素の中で最も軽量なものであり、非常に低い沸点と融点を持っています。このため、ヘリウムは常温・常圧下で気体として存在し、液体ヘリウムを得るには極低温下での処理が必要です。液体ヘリウムは熱伝導率が非常に高いです。
ヘリウムは非常に不活性な元素であり、化学的な反応性はほとんどありません。そのため、不活性ガスとして利用できます。
解説
ヘリウム,He.原子番号2の元素.電子配置1s2の周期表18族希ガス元素の一つ.原子量4.002602(2).天然に存在する同位体は 3He(0.000137(3)%),4He(99.999863(3)%).このほか,質量数5~10の放射性核種が報告されている.1868年にP.J.C. Janssenにより,日食の際の太陽スペクトル中に輝線(587.49 nm)として観測され,J.N. LockyerとE. Frankland(フランクランド)がギリシア語の太陽Ηλιο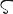 "(hèlios)にちなんでヘリウムと命名した.単体の分離精製は,1895年にW. Ramsay(ラムゼー)により,ウラン鉱物の一つ,クレーブ石(cleveite)から行われた.地球上では天然ガス中,あるいはα崩壊の産物としてウラン鉱物中に微量含まれ,体積で地球の大気の0.000524(5)% を占める.金,銀よりも希少な元素であるが,全宇宙には水素(74質量%)についで大量(26質量%)に存在する.工業的には,テキサス,カンザス,オクラホマなどから産出する天然ガスの液化と分留の繰り返しで得られる.埋蔵量は天然ガス埋蔵量と関係があるが,ヘリウム濃度の高低があるため,確認埋蔵量は全量の約1/4がカタール,それぞれ1/5がアルジェリアとアメリカ,ついでロシア.無色,無臭の気体.沸点4.230 K.臨界定数5.1953 K,0.22746 MPa,69.641 g cm-3.液体 4He は2.173 K(λ点)以下で超流動性を示す([別用語参照]液体ヘリウム).0.0025 K で 3He も超流動状態になる.常圧では0 K 付近でも固化しない唯一の例であるが,0.95 K で26 atm 加えれば固化する.固相も加圧により体積が容易に約30% もかわるなど特殊な性質を示す.第一イオン化エネルギー24.588 eV.化学的にはきわめて不活性で,通常の意味の化合物はつくらない.アーク溶接や半導体用高純度材料などの製造の際の不活性雰囲気として純ヘリウムが,深海作業あるいは加圧下の作業時にはHe 80%,O220% の混合気体が用いられる.医療用超伝導MRIやスペースシャトル打ち上げの際にも大量に使用される超低温用の冷媒としても重要である.近年,半導体だけでなく,液晶テレビや光ファイバー製造工程の不活性雰囲気,プラズマテレビの放電ガスとしても使用される重要な産業用ガスで,世界的に供給が不足している.アメリカはテキサス州に地下貯蔵施設をもち,大量のヘリウムガスを備蓄している.ロシアも東シベリア油田地区に貯蔵施設建設を計画している.
"(hèlios)にちなんでヘリウムと命名した.単体の分離精製は,1895年にW. Ramsay(ラムゼー)により,ウラン鉱物の一つ,クレーブ石(cleveite)から行われた.地球上では天然ガス中,あるいはα崩壊の産物としてウラン鉱物中に微量含まれ,体積で地球の大気の0.000524(5)% を占める.金,銀よりも希少な元素であるが,全宇宙には水素(74質量%)についで大量(26質量%)に存在する.工業的には,テキサス,カンザス,オクラホマなどから産出する天然ガスの液化と分留の繰り返しで得られる.埋蔵量は天然ガス埋蔵量と関係があるが,ヘリウム濃度の高低があるため,確認埋蔵量は全量の約1/4がカタール,それぞれ1/5がアルジェリアとアメリカ,ついでロシア.無色,無臭の気体.沸点4.230 K.臨界定数5.1953 K,0.22746 MPa,69.641 g cm-3.液体 4He は2.173 K(λ点)以下で超流動性を示す([別用語参照]液体ヘリウム).0.0025 K で 3He も超流動状態になる.常圧では0 K 付近でも固化しない唯一の例であるが,0.95 K で26 atm 加えれば固化する.固相も加圧により体積が容易に約30% もかわるなど特殊な性質を示す.第一イオン化エネルギー24.588 eV.化学的にはきわめて不活性で,通常の意味の化合物はつくらない.アーク溶接や半導体用高純度材料などの製造の際の不活性雰囲気として純ヘリウムが,深海作業あるいは加圧下の作業時にはHe 80%,O220% の混合気体が用いられる.医療用超伝導MRIやスペースシャトル打ち上げの際にも大量に使用される超低温用の冷媒としても重要である.近年,半導体だけでなく,液晶テレビや光ファイバー製造工程の不活性雰囲気,プラズマテレビの放電ガスとしても使用される重要な産業用ガスで,世界的に供給が不足している.アメリカはテキサス州に地下貯蔵施設をもち,大量のヘリウムガスを備蓄している.ロシアも東シベリア油田地区に貯蔵施設建設を計画している.
用途
液体ヘリウムはMRI装置内で低温発生のための寒冷剤、 セミコンダクター生産工程のガス、真空装置の漏洩検出装置、INE-HE レーザー、プラズマ装置内で使用されるガス混合物、Iガイガーカウンター内のネオン又はアルゴンとの混合ガス、ガス?タングステンアーク溶接におけるシールドガス、 高温の核反応装置内の冷媒、ガスクロ的分世紀装置のキャリヤーガス、不活性な環境を作るための不活性ガス、気球や飛行船の封入ガス
効能
医療用ガス (希釈剤)
説明
Helium is a colorless, odorless, and tasteless monoatomic gas with
an atomic weight of 4, a density of 1.78 g per 1 at 0°C and 1
atmosphere pressure, and an aqueous solubility of 0.97 mL per
100 mL at 50°C. The Bunsen solubility coefficient (the volume,
in mL, of gas at standard conditions dissolved per mL of liquid).
of helium at 38°C is 0.0086 in water, and 0.015 in olive oil. After
hydrogen, helium is the most abundant element in the universe;
these elements are believed to represent 76 and 23 percent, respectively, of all matter in the universe. The earth’s atmosphere, however, contains only 5 ppm of helium. Mineral gases from wells
contain higher concentrations and serve as the principal source for
industrial production. Helium is chemically very inert; it has a
weak tendency to combine with other elements such as fluorine.
It is usually marketed as compressed gas, with the USP grade
containing not less than 99 percent helium.
The largest biological use of helium is the replacement for nitrogen
in synthetic breathing gas mixtures for divers and others working
under high pressure conditions. The low solubility in body fluids
compared to nitrogen decreases the danger of bends on depressurizing.
化学的特性
Helium is a colorless, odorless, and tasteless
gas. It is nonflammable.
物理的性質
Helium is a colorless, odorless, and tasteless inert gas that is noncombustible and is the leastsoluble of any gas in water and alcohol. As a gas, it diffuses well in solids. Helium’s freezingpoint is –272.2°C, and its boiling point is –268.93°C. Both temperatures are near absolutezero (–273.13°C, or –459.4°F), where all molecular and thermal motion ceases. Liquid heliumhas the lowest temperature of any known substance. Helium’s density is 0.0001785g/cm
3.
Helium is the only element that cannot be converted into a solid by lowering the temperature.At normal pressure it remains a liquid at near absolute zero, but if the pressure isincreased, it then turns into a solid.
同位体
There are eight isotopes of helium. Two of these are stable. They are He-3,which makes up just 0.000137% of natural helium found on Earth, and He-4, whichaccounts for 99.999863% of the natural abundance of helium on Earth. Another isotope,He-5, is an extremely rare radioisotope that decays by emitting beta particles toform lithium-6 and lithium-8.
名前の由来
From the Greek word helios, meaning the “sun.” Through the process
of spectrometry, it was discovered on the sun before it was found on Earth in 1868.
特性
When a second proton and two neutrons are added to a hydrogen nucleus, a helium atomcan form after it collects two electrons. Helium is the most inert of all the noble group 18gases. It is so inactive that it does not even combine with itself. As a gas, helium remains asa single atom. The nuclei of helium are called alpha particles, each of which has a charge of+2 and an atomic mass of 4.
No stable compound of helium has ever been found. However, it is possible for an atom ofhydrogen to combine with helium (and other light noble elements) under special conditionsto form HeH+, an unstable ion.
Helium is not plentiful on Earth and is only the sixth most abundant gas in the atmosphere.It does not accumulate in the atmosphere because it is lighter than air. Some amountof helium continually escapes into space from the outer atmosphere of the Earth.
Liquid helium exhibits some unusual characteristics when supercooled. First, it is the onlyelement that will not turn into a solid by just using pressure. Heat must be removed as thepressure is increased, but helium will freeze at –272.2°C, which is the lowest temperature scientistshave ever achieved. Second, it is an excellent conductor of heat. As a supercold liquid,it will move toward heat—even flow up the sides and over the top of a container.
使用
Helium is used as an inert gas shield in arc
welding, as a lifting gas for lighter-than-air aircraft,
and as a gaseous cooling medium in nuclear
reactors. It is also used to provide a protective
atmosphere for growing germanium and
silicon crystals for transistors, to provide a protective
atmosphere in the production of such
reactive metals as titanium and zirconium, to fill
cold-weather fluorescent lamps, to trace leaks in
refrigeration and other closed systems, and to fill neutron and gas thermometers. Its thermal
conductivity makes it an important gas for lasers
and fiber optic production. It is used extensively
in chromatography due to its inertness and high
thermal conductivity. It is used in cryogenic
research such as for superconductivity. In mixtures
with oxygen, it has medical and diving
applications. Radioactive mixtures of helium
with krypton are available to users licensed by
the Nuclear Regulatory Commission. NASA
uses helium for purging and pressurizing the
liquid hydrogen tanks of spacecraft because it is
the only element that remains a gas in the extreme
cold necessary to maintain the liquid hydrogen
fuel used in many rockets and the Space
Shuttle. Liquid helium is used for cooling superconductive
magnets, used in magnetic resonance
imaging, and in magnetic separation.
調製方法
Helium is produced mostly by extraction from natural gas. The process involves cooling the stream of natural gas at sufficient low temperatures and high pressures to liquefy and separate all hydrocarbons, nitrogen and other gases from gaseous helium. The ‘crude’ helium gas may be purified further by repeated liquefaction of methane, nitrogen and other impurities under pressure. Trace hydrocarbons may be removed by adsorption on activated charcoal at liquid nitrogen temperature. Water may be removed by drying over a dehydrating agent such as bauxite. Carbon dioxide may be removed by passing helium through a scrubbing solution containing monoethanolamine-ethylene glycol or similar substances. Trace hydrogen may be removed from helium by converting it into water by mixing with oxygen and passing the mix over a palladium catalyst. Final purification may be achieved by adsorbing remaining trace contaminants over activated charcoal at liquid nitrogen temperature.
Alternatively, helium may be separated from natural gas by diffusion through permeable barriers, such as high silica glass or semipermeable membranes. The gas is supplied commercially in steel cylinders or tanks. The United States is the largest producer of helium in the world.
定義
Noble element of atomic number 2, first element
in the noble gas group of the periodic table, aw
4.00260, valence of 0. Helium nuclei are alpha particles. Most important isotope is helium-3.
一般的な説明
A colorless, odorless, noncombustible gas. Can asphyxiate. Inhalation causes the voice to become squeaky (Mickey Mouse voice). Exposure of the container to prolonged heat or fire can cause HELIUM to rupture violently and rocket. If liquefied, contact of the very cold liquid with water causes violent boiling. Pressures may build to dangerous levels if the liquid contacts water in a closed container. Used in arc welding, to trace leaks in refrigeration and other closed systems and as a lifting gas for lighter-than-air aircraft.
反応プロフィール
Chemically inert. These substances undergo no chemical reactions under any known circumstances. They are nonflammable, noncombustible and nontoxic. They can asphyxiate.
危険性
Being inert, the noble gases are nontoxic. However, they can act as asphyxiant gases thatcan kill because of oxygen deprivations.
A possible hazard is when He++ nuclei, as alpha particles, are accelerated to high speeds andbombard a target. Alpha particles can be stopped by several inches of air or a piece of cardboard.As high-energy, charged particles generated from man-made or natural radioactivity,alpha particles can cause damage, but they are not as damaging to our bodies as are very shortwavelength gamma rays, which can only be stopped by lead shielding.
健康ハザード
Vapors may cause dizziness or asphyxiation without warning. Vapors from liquefied gas are initially heavier than air and spread along ground.
火災危険
Non-flammable gases. Containers may explode when heated. Ruptured cylinders may rocket.
使用用途
ヘリウムの主な使用用途は下記の通りです。
(1)気球や飛行船の浮揚ガス
(2)半導体製造
(3)冷却剤(4)漏洩検査(5)光ファイバー(6)溶接(7)化学分析です
ソースです
ヘリウムは、大気中に体積で0.0005%程度しか含まれていませんが、アメリカなど一部の地域から採れる天然ガス中に多量に含まれています。その天然ガスからの分離・精製が主な生産方法です。 大気中のヘリウムの含有量はわずかであるため、窒素や酸素のように空気からヘリウムを分離することは工業的には行われていません。
工業用途
Helium is a colorless, odorless, elementary gas,He, with a specific gravity of 0.1368, liquefyingat –268.9°C, freezing at –272.2°C. It has avalency of zero and forms no electron-bondedcompounds. It has the highest ionization potentialof any element. The lifting power of heliumis only 92% that of hydrogen, but it is preferredfor balloons because it is inert and nonflammable,and is used in weather balloons. It is alsoused instead of air to inflate large tires for aircraftto save weight. Because of its low density,it is also used for diluting oxygen in the treatmentof respiratory diseases. Its heat conductivityis about six times that of air, and it is usedas a shielding gas in welding, and in vacuumtube and electric lamps. Because of its inertnesshelium can also be used to hold free chemicalradicals, which, when released, give highenergy and thrust for missile propulsion. Whenan electric current is passed through helium itgives a pinkish-violet light, and is thus used inadvertising signs. Helium can be obtained fromatmospheric nitrogen, but comes chiefly fromnatural gas.
材料の用途
Gaseous helium is noncorrosive and inert, and
may consequently be contained in systems constructed of any common metals and designed to
safely withstand the pressures involved. At the
temperature of liquid helium, ordinary carbon
steels and most alloy steels lose their ductility
and are considered unsafe for liquid helium
service. Satisfactory materials for use with liquid
helium include Type 18-8 stainless steel and
other austenitic nickel-chromium alloys, copper,
Monel, brass, and aluminum.
職業ばく露
It is used in weather balloons and in
welding gases. Liquid helium is used as a closed system
cooling agent.
貯蔵
Gaseous helium is commonly stored in high
pressure cylinders, hydril tubes, or tube trailers.
Liquid helium is commonly stored at the consumer
site in cryogenic liquid cylinders, portable
customer stations, and specially designed
insulated tanks. To minimize helium transfer losses, the shipping container for liquid helium
is normally used for storage.
輸送方法
UN1046 Helium, compressed, Hazard Class: 2.2;
Labels: 2.2-Nonflammable compressed gas; UN1963
Helium, refrigerated liquid (cryogenic liquid), Hazard
Class: 2.2; Labels: 2.2-Nonflammable compressed gas.
純化方法
Dry the gas by passing it through a column of Linde 5A molecular sieves and CaSO4, then through an activated-charcoal trap cooled in liquid N2, to adsorb N2, argon, xenon and krypton. Also pass it over CuO pellets at 300o to remove hydrogen and hydrocarbons, over Ca chips at 600o to remove oxygen, and then over titanium chips at 700o to remove N2 [Arnold & Smith J Chem Soc, Faraday Trans 2 77 861 1981]. Its solubility in 100mL of H2O is 0.94mL at 25o, 1.05mL at 50o and 1.21 at 75o.
廃棄物の処理
Return refillable compressed
gas cylinders to supplier.
ヘリウム 上流と下流の製品情報
原材料
準備製品