L-(-)-페닐알라닌
|
|
L-(-)-페닐알라닌 속성
- 녹는점
- 270-275 °C (dec.)(lit.)
- 알파
- -34.1 º (c=2, water, dry basis)
- 끓는 점
- 293.03°C (rough estimate)
- 밀도
- 1.29
- 증기압
- <1 Pa (25 °C)
- 굴절률
- -34 ° (C=2, H2O)
- 저장 조건
- Store below +30°C.
- 용해도
- H2O: 0.1 M at 20 °C, 투명, 무색
- 물리적 상태
- 가루
- 산도 계수 (pKa)
- 2.2(at 25℃)
- 색상
- 흰색에서 황백색까지
- 수소이온지수(pH)
- 5.0-7.0 (25℃, 0.1M in H2O)
- 냄새
- 100.00%에서. 냄새 없는
- ?? ??
- 냄새 없는
- optical activity
- [α]25/D -34.2°, c = 2 in H2O (dried basis)
- 수용성
- 1~5g/100mL, 25 ºC
- JECFA Number
- 1428
- Merck
- 14,7271
- BRN
- 1910408
- 안정성
- 안정적인. 강한 산화제와 호환되지 않습니다.
- InChIKey
- COLNVLDHVKWLRT-QMMMGPOBSA-N
- LogP
- 0.24
- CAS 데이터베이스
- 63-91-2(CAS DataBase Reference)
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | C | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38-34 | ||
| 안전지침서 | 22-24/25-37/39-45-36/37/39-27-26 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | AY7535000 | ||
| F 고인화성물질 | 10 | ||
| TSCA | Yes | ||
| HS 번호 | 29224995 | ||
| 유해 물질 데이터 | 63-91-2(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-01557 |
L-(-)-페닐알라닌 C화학적 특성, 용도, 생산
순도시험
(1) 용상 : 이 품목 1g을 물 100mL에 녹일 때, 그 액은 무색으로서 탁도는 거의 징명 이하이어야 한다.
(2) 액성 : 이 품목의 수용액(1→100)의 pH는 5.4~6.0이어야 한다.
(3) 비선광도 : 이 품목을 105℃에서 3시간 건조한 다음 약 1g을 정밀히 달아 물에 녹여 50mL로 하여 이 액의 선광도를 측정할 때, 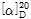 =-33.0~-35.2°이어야 한다.
=-33.0~-35.2°이어야 한다.
(4) 비소 : 이 품목을 비소시험법에 따라 시험할 때, 그 양은 4.0ppm 이하이어야 한다.
(5) 납 : 이 품목 5.0g을 취하여 원자흡광광도법 또는 유도결합플라즈마발광광도법에 따라 시험할 때, 그 양은 5.0ppm 이하이어야 한다.
(6) 염화물 : 이 품목 0.5g을 취하여 염화물시험법에 따라 시험할 때, 그 양은 0.01N 염산 0.3mL에 대응하는 양 이하이어야 한다.
확인시험
(1) 이 품목의 수용액(1→1,000) 5mL에 중크롬산칼륨용액(1→1,000) 1mL를 가하여 끓이면 특유한 냄새가 발생한다.
(2) 이 품목의 수용액(1→1,000) 5mL에 닌히드린시액 1mL를 가하여 3분간 가열하면 적자~청자색을 나타낸다.
(3) 이 품목 10mg에 질산칼륨 0.5g 및 황산 2mL를 가하여 수욕상에서 20분간 가열한 다음 식히고 염산히드록실아민용액(1→10) 5mL를 가하여 얼음물 중에 10분간 방치한 다음 즉시 40% 수산화나트륨용액 8mL를 가하여 방치하면 적자색을 나타낸다.
정량법
이 품목을 건조한 다음 약 0.3g을 정밀히 달아 이하 「글리신」의 정량법에 따라 정량한다.
0.1N 과염소산용액 1mL = 16.52mg C9H11O2N
강열잔류물
이 품목의 강열잔류물은 0.1% 이하이어야 한다.
화학적 성질
L-Phenylalanine has no odor and a slight bitter taste. It melts with decomposition at about 283°C. The pH of a 1 in 100 solution is between 5.4 and 6.0. FEMA notes this chemical is used in cocoa substitute only.출처
Reported found in white bread, macaroni, egg noodles, corn flakes, corn grits, oatmeal, wheat bran, wheat flakes, shredded wheat, barley, brown rice, rye flour, whole grain wheat flour, buttermilk, blue cheese, cheddar cheese, cottage cheese, cream cheese, parmesan cheese, bacon, cured ham, frankfurters, pork sausage, canned red kidney beans, canned sweet corn, canned peas, canned lima beans, canned potatoes, canned asparagus, canned snap beans, canned beets, beef, lamb, fresh ham, veal round, beef liver, chicken, chicken liver, turkey and other natural sources.용도
L-Phenylalanine is an essential amino acid. L-Phenylalanine is biologically converted into L-tyrosine, another one of the DNA-encoded amino acids, which in turn is converted to L-DOPA and further conv erted into dopamine, norepinephrine, and epinephrine. L-Phenylalanine is produced for medical, feed, and nutritional applications such as in the preparation of Aspartame.생산 방법
In previous large-scale production processes for L-phenylalanine two enzymatic methods were applied: 1. Resolution of N-acetyl-D,L-phenylalanine by carrier-fixed microbial acylase: This process provided pharmaceutical-grade L-phenylalanine, but suffered from the disadvantage that the D-enantiomer had to be racemized and recycled. 2. Stereoselective and enantioselective addition of ammonia to trans-cinnamic acid, catalyzed by L-phenylalanine ammonia lyase (PAL, EC 4.3.1.5): PAL-containing Rhodotorula rubra was used in an industrial process to supply L-phenylalanine for the first production campaign of the sweetener aspartame. When continuously operated in an immobilized whole cell reactor, the bioconversion reached concentration up to 50 g/L Lphenylalanine at a conversion of about 83%. Other processes started from phenylpyruvate with L-aspartic acid as amine donor using immobilized cells of Escherichia coli or from a-acetamidocinnamic acid and immobilized cells of a Corynebacterium equi strain. In both cases L-phenylalanine concentrations up to 30 g/L and more (molar yields as high as at least 98 %) were reached.However, fermentation processes based on glucose-consuming L-phenylalanine overproducing ;mutants of E. coli and coryneform strains turned out to be more economical. L-Phenylalanine is formed in ten enzymatic steps starting from erythrose-4-phosphate and phosphoenolpyruvate. A suitable profile of the specific glucose feed rate prevents acetate formation and leads to improved L-phenylalanine production with a final concentration up to 46 g/L and a corresponding yield of 18 %.
L-Phenylalanine is recovered from the fermentation broth either by two-step crystallization or by an ionexchange resin process. The preferred cell separation technique is ultrafiltration; and the filtrates may be treated with activated carbon for further purification. Instead of ion-exchange resins nonpolar, highly porous synthetic adsorbents are recommended to remove impurities. An alternative process in which a cell separator is integrated in the fermentation part, thus allowing cell recycling, was suggested for L-phenylalanine production and may lead to prospective developments.
정의
ChEBI: The L-enantiomer of phenylalanine.제조 방법
From PTS-negative Escherichia coli bioengineered strains.일반 설명
Odorless white crystalline powder. Slightly bitter taste. pH (1% aqueous solution) 5.4 to 6.공기와 물의 반응
Water soluble. Aqueous solutions are weak acids.반응 프로필
L-Phenylalanine may be light sensitive. Act as weak acids in solution.건강위험
ACUTE/CHRONIC HAZARDS: When heated to decomposition L-Phenylalanine emits toxic fumes of nitrogen oxides.화재위험
Flash point data for L-Phenylalanine are not available; however, L-Phenylalanine is probably combustible.Safety Profile
Mildly toxic by intraperitoneal route. An experimental teratogen. Experimental reproductive effects. Human mutation data reported. When heated to decomposition it emits toxic fumes of NOx.Purification Methods
Likely impurities are leucine, valine, methionine and tyrosine. Crystallise L-phenylalanine from water by adding 4volumes of EtOH. Dry it in vacuo over P2O5. Also crystallise it from saturated refluxing aqueous solutions at neutral pH, or 1:1 (v/v) EtOH/water solution, or conc HCl. It sublimes at 176-184o/0.3mm with 98.7% recovery and unracemised [Gross & Gradsky J Am Chem Soc 77 1678 1955]. [Greenstein & Winitz The Chemistry of the Amino Acids J. Wiley, Vol 3 pp 2156-2175 1961, Beilstein 14 IV 1552.]L-(-)-페닐알라닌 준비 용품 및 원자재
원자재
준비 용품
L-(-)-페닐알라닌 공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Longbang Technology Co., LTD | +86-18032476855 +86-18032476855 |
admin@hblongbang.com | China | 902 | 58 |
| Hebei Kingfiner Technology Development Co.Ltd | +86-15532196582 +86-15373005021 |
lisa@kingfinertech.com | China | 3002 | 58 |
| Guangzhou Tengyue Chemical Co., Ltd. | +86-86-18148706580 +8618826483838 |
evan@tyvovo.com | China | 154 | 58 |
| Shaanxi Haibo Biotechnology Co., Ltd | +undefined18602966907 |
qinhe02@xaltbio.com | China | 1000 | 58 |
| Anhui Ruihan Technology Co., Ltd | +8617756083858 |
daisy@anhuiruihan.com | China | 994 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 18219 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-18034520335 |
admin@hbsaisier.cn | China | 940 | 58 |
| Ouhuang Engineering Materials (Hubei) Co., Ltd | +8617702722807 |
admin@hbouhuang.com | China | 3001 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 |
admin@zlchemi.com | China | 2603 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |







