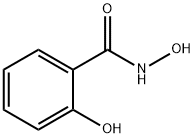
Salicylhydroxamic acid
|
|
Salicylhydroxamic acid 속성
- 녹는점
- 177 °C (dec.) (lit.)
- 끓는 점
- 276.03°C (rough estimate)
- 밀도
- 1.3585 (rough estimate)
- 굴절률
- 1.5500 (estimate)
- 저장 조건
- Sealed in dry,Room Temperature
- 물리적 상태
- 미세 결정성 분말
- 산도 계수 (pKa)
- pK (25°) 4.19
- 색상
- 베이지 핑크
- 수용성
- 물에 용해됩니다.
- Merck
- 14,8331
- BRN
- 1210520
- 안정성
- 안정적인. 강염기, 강산화제와 호환되지 않습니다. 타기 쉬운.
- CAS 데이터베이스
- 89-73-6(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 20/21/22-36/37/38-40 | ||
| 안전지침서 | 22-26-36-24/25 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | VO6870000 | ||
| HS 번호 | 29242990 |
Salicylhydroxamic acid C화학적 특성, 용도, 생산
화학적 성질
It is a white needle crystal or off-white powder with a slow-heating melting point of 168°C. It is soluble in alcohol and ether and can be sublimated. When exposed to air, it progressively turns red. It is synthesized by combining methyl salicylate and hydroxylamine hydrochloride with sodium hydroxide, followed by acidification.용도
Salicylhydroxamic acid is a collector with efficient chelating effect on rare metal oxide minerals. It is used in rare metal beneficiation and has the advantages of good selectivity and strong collecting power. Salicylhydroxamic acid is mainly used as a complexing agent or extractant for rare earth ore, copper oxide ore, lead-zinc oxide ore, gold ore, kaolin, etc., as well as organic synthesis intermediates.제조 방법
The synthesis of salicylhydroxamic acid involved a two-step process. In the first step, methyl salicylate was prepared by esterifying salicylic acid with methanol using 28g (0.2 mol) of salicylic acid, 81 ml of methanol, and 8 ml of concentrated sulfuric acid. In the second step, hydroxylamine was coupled with the methyl salicylate. 13.9 g (0.2mol) of hydroxylamine was mixed with 200 ml of 10% sodium hydroxide solution and cooled. To this, 152.3g (0.1mol) of methyl salicylate was added in small portions with vigorous shaking after each addition to ensure complete dissolution. The mixture was allowed to stand for 36 hours, then acidified with 2M sulphuric acid and cooled. The precipitate was filtered, recrystallized from hot water containing a drop of acetic acid, recooled, and filtered. The yield of white precipitate collected and weighed was 14g of salicylhydroxamic acid.Preparation and Characterization of Salicylhydroxamic acid and its Complexes with Iron (III), Cobalt (II) and Manganese (II)
정의
ChEBI: Salicylhydroxamic acid is a hydroxamic acid that is N-hydroxybenzamide carrying a phenolic hydroxy group at position 2. It has a role as an antibacterial drug, a trypanocidal drug, an EC 3.5.1.5 (urease) inhibitor and an EC 1.11.2.2 (myeloperoxidase) inhibitor. It is a member of phenols and a hydroxamic acid.Purification Methods
Crystallise the hydroxamic acid from acetic acid. [Beilstein 10 H 98.]Salicylhydroxamic acid 준비 용품 및 원자재
원자재
준비 용품
Salicylhydroxamic acid 공급 업체
글로벌( 261)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12470 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +86-18949832763 |
info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8829 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 |
sales@alchempharmtech.com | United States | 63711 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 15319487004 |
1015@dideu.com | China | 2263 | 58 |
| Xingtai Haoxun Import and Export Trade Co., Ltd. | +8617733976525 |
lisa@xthaoxun.com | CHINA | 258 | 58 |
Salicylhydroxamic acid 관련 검색:
살리실알독심 아세토히드록삼 산 엽산 글리신 시트르산 아세트산
Alkyl hydroxy oximido acid
Benzohydroxamic acid
Ascoric Acid
2-(2-CHLOROETHYL)-3-HYDROXY-3,4-DIHYDRO-2H-1,3-BENZOXAZIN-4-ONE
N-METHOXY-2,5-BIS(2,2,2-TRIFLUOROETHOXY)BENZENECARBOXAMIDE
Salicylhydroxamic acid
phosphoric acid
2-(2-CHLOROETHYL)-4-OXO-3,4-DIHYDRO-2H-1,3-BENZOXAZIN-3-YL N-PHENYLCARBAMATE
2-(2-CHLOROETHYL)-4-OXO-3,4-DIHYDRO-2H-1,3-BENZOXAZIN-3-YL N-(4-CHLOROPHENYL)CARBAMATE
N-[(3,4-DICHLOROBENZYL)OXY]-2,5-BIS(2,2,2-TRIFLUOROETHOXY)BENZENECARBOXAMIDE
2-(([2,5-BIS(2,2,2-TRIFLUOROETHOXY)BENZOYL]AMINO)OXY)ACETIC ACID
N-HYDROXY-2,5-BIS(2,2,2-TRIFLUOROETHOXY)BENZENECARBOXAMIDE