Capmatinib hydrochloride manufacturers
|
| | Capmatinib hydrochloride Basic information |
| Product Name: | Capmatinib hydrochloride | | Synonyms: | Capmatinib hydrochloride;Capmatinib hydrochloride Hydrate;Capmatinib HCl hydrate (INCB-28060;Capmatinib 2HCl.H2O;INC-280 2HCl.H2O;INCB28060 2HCl.H2O;NVP-INC280 2HCl.H2O;Capmatinib Hydrochloride Hydrate
(INCB28060) | | CAS: | 1865733-40-9 | | MF: | C23H20ClFN6O2 | | MW: | 466.9 | | EINECS: | | | Product Categories: | | | Mol File: | 1865733-40-9.mol | 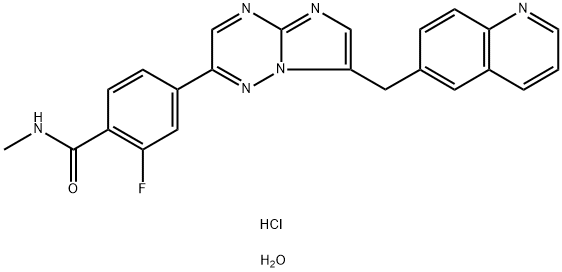 |
| | Capmatinib hydrochloride Chemical Properties |
| InChIKey | XCHIKTXPDQTRMI-UHFFFAOYSA-N | | SMILES | C(C1C=CC2N=CC=CC=2C=1)C1=CN=C2N=CC(C3C=CC(C(=O)NC)=C(F)C=3)=NN12.Cl.O |
| | Capmatinib hydrochloride Usage And Synthesis |
| Description | Capmatinib dihydrochloride hydrate is a orally active,reversible MET tyrosine kinase inhibitor that received approval for advanced non-small cell lung cancer(NSCLC) harboring MET exon 14 skipping mutationa. Capmatinib dihydrochloride hydrate can inhibit phosphorylation of c-MET as well as c-MET pathway downstream effectors such as ERK1/2, AKT, FAK, GAB1, and STAT3/5. Capmatinib dihydrochloride hydrate potently inhibits c-MET-dependent tumor cell proliferation and migration and effectively induces apoptosis. Antitumor activity. Capmatinib dihydrochloride hydrate is largely metabolized by CYP3A4 and aldehyde oxidase[1-3]. | | Origin | Capmatinib was first reported in 2011 by Liu et al., who showed that in both in vivo and in vitro mice studies using human cell lines, capmatinib had a 10,000-fold selectivity for c-met over a large panel of human kinase. They showed that capmatinib can block the c-MET phosphorylation and activation of downstream targets, including HGF. They further showed that activated c-met upregulates cancer-promoting EGFR and HER-3 pathways. Baltschukat et al. further investigated capmatinib in NSCLC[4]. | | Mechanism of action | Tabrecta (capmatinib) is a kinase inhibitor that targets MET, including the mutant variant produced by exon 14 skipping. MET exon 14 skipping results in a protein with a missing regulatory domain that reduces its negative regulation, leading to increased downstream MET signaling. | | Toxicity evaluation | Effects During Pregnancy and Lactation No information is available on the clinical use of capmatinib during breastfeeding. Because capmatinib is 96% bound to plasma proteins, the amount in milk is likely to be low. The manufacturer recommends that breastfeeding be discontinued during capmatinib therapy and for 1 week after the last dose.
| | References | [1] HSUROBERT NagasakaMisako BenjaminDavid J. The Development and Role of Capmatinib in the Treatment of MET-Dysregulated Non-Small Cell Lung Cancer-A Narrative Review.[J]. Cancers, 2023. DOI:10.3390/cancers15143561.
[2] LIUXIANGDONG. A novel kinase inhibitor, INCB28060, blocks c-MET-dependent signaling, neoplastic activities, and cross-talk with EGFR and HER-3.[J]. Clinical Cancer Research, 2011. DOI:10.1158/1078-0432.CCR-11-1157.
[3] BALTSCHUKATSABRINA. Capmatinib (INC280) Is Active Against Models of Non-Small Cell Lung Cancer and Other Cancer Types with Defined Mechanisms of MET Activation.[J]. Clinical Cancer Research, 2019. DOI:10.1158/1078-0432.CCR-18-2814.
[4] HsuRobert, NagasakaMisako, BenjaminDavid J. "The Development and Role of Capmatinib in the Treatment of MET-Dysregulated Non-Small Cell Lung Cancer-A Narrative Review." Cancers 15 14 (2023). |
| | Capmatinib hydrochloride Preparation Products And Raw materials |
|