Upadacitinib hemihydrate manufacturers
|
| | Upadacitinib hemihydrate Basic information |
| Product Name: | Upadacitinib hemihydrate | | Synonyms: | 1-Pyrrolidinecarboxamide, 3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)-, hydrate (2:1), (3S,4R)-;Upadacitinib hemihydrate;Upadacitinib Hydrate;Cinchonanium, 1-[13,5-bis(trifluoromethyllphenyllmethyl]-9-hydroxy-1'-
[[3-(trifluoromethyl)phenyllmethyl},bromide(1:2), (8a,9R)-";ABT 494 hemihydrate;ABT494 hemihydrate;ABT-494 hemihydrate | | CAS: | 2050057-56-0 | | MF: | C17H21F3N6O2 | | MW: | 398.39 | | EINECS: | | | Product Categories: | API | | Mol File: | 2050057-56-0.mol | 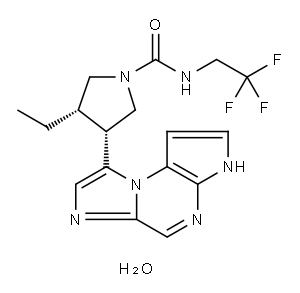 |
| | Upadacitinib hemihydrate Chemical Properties |
| InChIKey | VJSRYIXHEWRBKD-NFGOAHBONA-N | | SMILES | C([C@@H]1CN(C(=O)NCC(F)(F)F)C[C@@H]1C1=CN=C2C=NC3NC=CC=3N12)C.O |&1:1,13,r| |
| | Upadacitinib hemihydrate Usage And Synthesis |
| Clinical Use | RINVOQ is indicated for the treatment of moderate to severe active rheumatoid arthritis in adult patients who have responded inadequately to, or who are intolerant to one or more disease-modifying anti-rheumatic drugs (DMARDs). RINVOQ may be used as monotherapy or in combination with methotrexate. Each prolonged-release tablet (RINVOQ 15 mg prolonged-release tablets.) contains upadacitinib hemihydrate, equivalent to 15 mg of upadacitinib. |
| | Upadacitinib hemihydrate Preparation Products And Raw materials |
|