|
|
| | [BIS(TRIMETHYLSILYL)]SELENIDE Basic information |
| | [BIS(TRIMETHYLSILYL)]SELENIDE Chemical Properties |
| Melting point | -7°C | | Boiling point | 58-9°C/11mmHg | | density | 0,9 g/cm3 | | refractive index | 1.481 | | Fp | 59.6±18.7℃ | | solubility | sol Et2O and THF. | | form | liquid | | Specific Gravity | 0.90 | | Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| | [BIS(TRIMETHYLSILYL)]SELENIDE Usage And Synthesis |
| Chemical Properties | CLEAR COLOURLESS TO YELLOW LIQUID | | Physical properties | bp 45–46°C/5.3 mmHg. | | Uses | Bis(trimethylsilyl) Selenide can be used as synthesis of unsymmetrical selenides; generation of selenoaldehydes;
reduction of sulfoxides, selenoxides, and
telluroxides).
Bis(trimethylsilyl)
selenide reacts with equimolar amounts of n-butyllithium to generate
Me3SiSeLi, alkylation of which then provides trimethylsilyl
alkyl selenides. Similar treatment of Me3SiSeR with BuLi /
R2X successfully leads to unsymmetrical selenides in good yields
(eq 5).Use of acid chlorides in place of alkyl halides results in
the formation of selenoesters.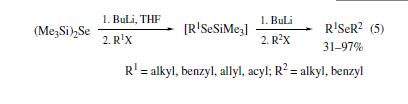 | | Preparation | In the initial studies,
bis(trimethylsilyl) selenide was synthesized by the following
two methods: silylation of sodium selenide (or lithium selenide)
with chlorotrimethylsilane (eq 1) or reaction of bromobenzene,
magnesium, and selenium with chlorotrimethylsilane (eq 2). 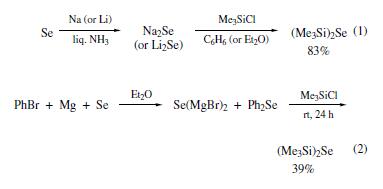
|
| | [BIS(TRIMETHYLSILYL)]SELENIDE Preparation Products And Raw materials |
|