| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:Nα-Fmoc-Nβ-(N-Boc-amino-oxyacetyl)-L-2,3-diaminopropionic acid, 98%
CAS:600153-12-6
Purity:98% Package:1G;250MG
|
|
| | FMOC-DPR(BOC-AOA)-OH Basic information |
| Product Name: | FMOC-DPR(BOC-AOA)-OH | | Synonyms: | FMOC-DAP[AC(-O-NHBOC)]-OH;FMOC-DAP(BOC-AOA)-OH;FMOC-DPR(BOC-AOA)-OH;FMOC-L-DAP(BOC-AOA);FMOC-L-DAP(BOC-AOA)-OH;FMOC-L-DAPA(BOC-AOA)-OH;N-ALPHA-(9-FLUORENYLMETHOXYCARBONYL)-N-BETA-(T-BUTOXYCARBONYL-AMINOOXYACETYL)-L-2,3-DIAMINOPROPIONIC ACID;N-ALPHA-(9-FLUORENYLMETHYLOXYCARBONYL)-N-BETA-(TERT-BUTYLOXYCARBONYL-AMINOOXY-ACETYL)-L-2,3-DIAMINOPROPIONIC ACID | | CAS: | 600153-12-6 | | MF: | C25H29N3O8 | | MW: | 499.51 | | EINECS: | | | Product Categories: | | | Mol File: | 600153-12-6.mol | 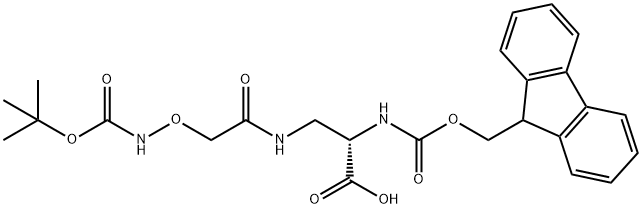 |
| | FMOC-DPR(BOC-AOA)-OH Chemical Properties |
| density | 1.304±0.06 g/cm3(Predicted) | | storage temp. | Store below +30°C. | | pka | 3.41±0.10(Predicted) |
| | FMOC-DPR(BOC-AOA)-OH Usage And Synthesis |
| Chemical Properties | White crystalline powder | | General Description | An excellent derivative for the chemoselective ligation of unprotected peptides in aqueous media [1] via oxime formation. This derivative introduces a hydroxylamine functionality which can couple with aldehyde groups present in another peptide unit.
The product number for this product was previously 04-12-1185.
To obtain a certificate of analysis (CoA) of a lot that begins with the letter “A”, please select the option in the right hand menu “Request a COA for Lot#s starting with A”. |
| | FMOC-DPR(BOC-AOA)-OH Preparation Products And Raw materials |
|