|
|
| | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol Basic information |
| Product Name: | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol | | Synonyms: | A,A-DIPHENYL-L-PROLINOL;(s)-(-)-à,à-diphenyl-2-pyrrolidinemethanol;(s)-(-)-à,à-diphenylprolinol;α,α-diphenyl-l-prolinol;2,2-DIPHENYL-(2S)-2-PYRROLIDINEMETHANOL;(S)-(-)-alpha,alpha-diphenyl-2-pyrrolidine;(S)-(-)-ALPHA ALPHA-DIPHENYL-2- PYRROLIDINEMETHANOL 99%;2-PYRROLIDINEMETHANOL, .ALPHA.,.ALPHA.-DIPHENYL-, (2S)- | | CAS: | 112068-01-6 | | MF: | C17H19NO | | MW: | 253.34 | | EINECS: | 601-153-1 | | Product Categories: | Peptide;Asymmetric Synthesis;Chiral Building Blocks;Simple Alcohols (Chiral);Synthetic Organic Chemistry;CHIRAL CHEMICALS;Chiral chemicals;Pharmaceutical intermediate;Chiral Nitrogen;Benzenes;Chiral Reagent;Chiral Reagents;chiral | | Mol File: | 112068-01-6.mol | 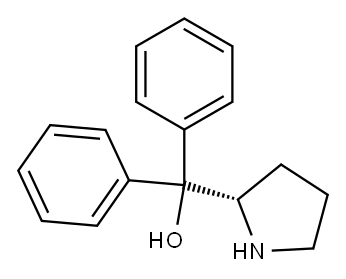 |
| | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol Chemical Properties |
| Melting point | 77-80 °C(lit.) | | alpha | -59 º (c=3, methanol 25 ºC) | | Boiling point | 396.54°C (rough estimate) | | density | 1.0078 (rough estimate) | | refractive index | -66.5 ° (C=3, CHCl3) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | Soluble in chloroform. | | form | powder | | pka | 13.15±0.29(Predicted) | | color | white to off white ., crystal | | optical activity | [α]20/D 67°, c = 3 in chloroform | | Merck | 14,155 | | BRN | 17103 | | InChIKey | OGCGXUGBDJGFFY-INIZCTEOSA-N | | CAS DataBase Reference | 112068-01-6(CAS DataBase Reference) | | NIST Chemistry Reference | S-(-)-1,1-diphenylprolinol(112068-01-6) |
| | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol Usage And Synthesis |
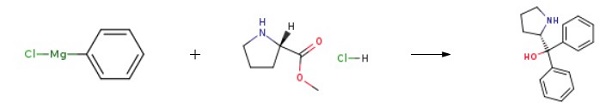
| Chemical Properties | white to beige crystals or crystalline powder | | Uses | suzuki reaction | | Uses | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol is used to prepare the corresponding oxazaborolidines for the borane-mediated asymmetric reduction of ketones. | | Preparation | Into a four-necked flask, add tetrahydrofuran, L-proline methyl ester hydrochloride, and phenyl magnesium chloride (dissolved in tetrahydrofuran) solution was added dropwise from a constant pressure dropping funnel, and the addition was completed in about 4 hours, and then stirred at 10-15°C for 3 hours. Add water, adjust the pH to 5-6 with 10% ammonium chloride aqueous solution, extract with dichloromethane, extract the aqueous layer twice with dichloromethane each time, and obtain the organic phase. Wash once with saturated sodium chloride aqueous solution. The organic phase was dried with anhydrous sodium sulfate for 4 hours and filtered. The obtained organic phase was distilled under reduced pressure to distill dichloromethane and tetrahydrofuran to obtain light yellow oily (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol, yield 86.6%.
 | | Application | (S)-(+)-α,α-Diphenyl-2-pyrrolidinemethanol (DPPM) may be used in the following processes:- DPPM
reacts with catecholborane to form a spiroborate ester, which can be an
efficient catalyst for the synthesis of enantiopure alcohols by borane
reduction of acetophenone.
- The
catalyst generated in situ by reacting DPPM with borane-diethylaniline,
can efficiently catalyze the enantioselective reduction of
2′-fluoroacetophenone.
- Mesoporous SBA-15 silica functionalized with DPPM can catalyze the addition of diethylzinc to benzaldehyde to form (S)-1-phenyl-propanol.
Used to prepare the corresponding oxazaborolidines for the borane-mediated asymmetric reduction of ketones. | | General Description | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol is a bifunctional organocatalyst. |
| | (S)-(-)-α,α-Diphenyl-2-pyrrolidinemethanol Preparation Products And Raw materials |
|