|
|
| | 5-AMino-2,4-di-tert-butylphenol Basic information |
| Product Name: | 5-AMino-2,4-di-tert-butylphenol | | Synonyms: | 5-AMino-2,4-di-tert-butylphenol;2,4-Di-tert-butyl-5-aMinophenyl Methyl carbonate;Phenol, 5-amino-2,4-bis(1,1-dimethylethyl)-;5-Amino-2,4-bis(2-methyl-2-propanyl)phenol;VX-770 intermediate;5-amino-2,4-di-tert-butylphenol hydrochloride (base);4,6-dichloropyridazin-4-ol;5-Amino-2,4-ditertiarybutyphenol | | CAS: | 873055-58-4 | | MF: | C14H23NO | | MW: | 221.34 | | EINECS: | | | Product Categories: | | | Mol File: | 873055-58-4.mol | 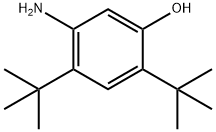 |
| | 5-AMino-2,4-di-tert-butylphenol Chemical Properties |
| Boiling point | 319.7±30.0 °C(Predicted) | | density | 0.990±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C(protect from light) | | pka | 11.63±0.23(Predicted) | | InChI | InChI=1S/C14H23NO/c1-13(2,3)9-7-10(14(4,5)6)12(16)8-11(9)15/h7-8,16H,15H2,1-6H3 | | InChIKey | BKSDHPHOWDUJNB-UHFFFAOYSA-N | | SMILES | C1(O)=CC(N)=C(C(C)(C)C)C=C1C(C)(C)C |
| | 5-AMino-2,4-di-tert-butylphenol Usage And Synthesis |
| Uses | 5-Amino-2,4-di-tert-butylphenol is an intermediate used in the preparation of ivacaftor via condensation of oxo dihydroquinoline carboxylic acid with amino ditert butylphenol. | | Synthesis | To a refluxing solution of 2,4-di-tert-butyl-5-nitro-phenol (1.86 g, 7.40 mmol) and ammonium formate (1.86 g) in ethanol (75 mL) was added Pd-5% wt on activated carbon (900 mg). The reaction mixture was stirred at reflux for 2 h, cooled to room temperature, and filtered through Celite. The Celite was washed with methanol, and the combined filtrates were concentrated to yield 5-amino-2,4-di-tert-butyl-phenol as a grey solid (1.66 g, quant.).
|
| | 5-AMino-2,4-di-tert-butylphenol Preparation Products And Raw materials |
|