- coumarin 460
-

- $30000.00 / 1T
-
2023-12-21
- CAS:91-44-1
- Min. Order: 1T
- Purity: 99%
- Supply Ability: 20tons
|
| | 7-Diethylamino-4-methylcoumarin Basic information |
| Product Name: | 7-Diethylamino-4-methylcoumarin | | Synonyms: | 7-DIETHYLAMINO-4-METHYLCOUMARIN;7-DIETHYLAMINO-4-METHYLCOUMARINE;Leucophor WS;BRIGHTENER 61;COUMARIN I;COUMARIN 1;c.i. fluorescent brightener 140;C.I. Fluorescent Brightener 52 | | CAS: | 91-44-1 | | MF: | C14H17NO2 | | MW: | 231.29 | | EINECS: | 202-068-9 | | Product Categories: | | | Mol File: | 91-44-1.mol | 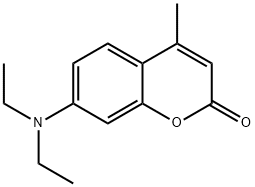 |
| | 7-Diethylamino-4-methylcoumarin Chemical Properties |
| Melting point | 72-75 °C (lit.) | | Boiling point | 240 °C / 6.5mmHg | | density | 1.122 | | vapor pressure | 0-0.003Pa at 20-25℃ | | refractive index | 1.5300 (estimate) | | Fp | 152°C | | storage temp. | Sealed in dry,Room Temperature | | solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 3.51±0.20(Predicted) | | form | Crystalline Powder | | color | Light beige to light purple | | Water Solubility | slightly soluble | | BRN | 193303 | | Stability: | Stable. Incompatible with strong oxidizing agents. | | InChIKey | AFYCEAFSNDLKSX-UHFFFAOYSA-N | | LogP | 2.523-2.8 at 23-25℃ | | CAS DataBase Reference | 91-44-1(CAS DataBase Reference) | | NIST Chemistry Reference | Coumarin, 7-(diethylamino)-4-methyl-(91-44-1) | | EPA Substance Registry System | 7-Diethylamino-4-methylcoumarin (91-44-1) |
| | 7-Diethylamino-4-methylcoumarin Usage And Synthesis |
| Chemical Properties | light tan powder | | Uses | 7-(Diethylamino)-4-methylcoumarin, is potentially useful as a laser dye. | | Uses | Optical bleach in textile industry, in coatings for paper, labels, book covers, etc.; to lighten plastics, resins, varnishes, and lacquers; invisible marking agent. | | Definition | ChEBI: Coumarin 460 is a member of 7-aminocoumarins. It has a role as a fluorochrome. | | General Description | Light tan grains. Gives bright blue-white fluorescence in very dilute solutions. | | Reactivity Profile | 7-Diethylamino-4-methylcoumarin is a lactone (behaves as an ester) and an amine. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. | | Flammability and Explosibility | Not classified |
| | 7-Diethylamino-4-methylcoumarin Preparation Products And Raw materials |
|