- TETRATHIAFULVALENE
-

- $3.00 / 1KG
-
2020-01-08
- CAS:31366-25-3
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 200KG
|
| | TETRATHIAFULVALENE Basic information |
| Product Name: | TETRATHIAFULVALENE | | Synonyms: | 1,3-Dithiole, 2-(1,3-dithiol-2-ylidene)-;1,4,5,8-Tetrathiafulvalen;1,4,5,8-Tetrathiafulvalene;2-(1,3-dithiol-2-ylidene)-1,3-dithiol;Δ2,2μ-Bi-1,3-dithiole, TTF;Delta2,2'-bi-1,3-dithiol;[2,2']-Bi[1,3]-dithioylidene;Tetrathiafulvalene, 99+% 1GR | | CAS: | 31366-25-3 | | MF: | C6H4S4 | | MW: | 204.34 | | EINECS: | 250-593-7 | | Product Categories: | Functional Materials;TTF Derivatives;Building Blocks;Chemical Synthesis;Heterocyclic Building Blocks;Materials Science;Organic and Printed Electronics;Organic Field Effect Transistor (OFET) Materials;Others;p-Type Organic Semiconductors;p-Type Small Molecules;S-Containing;Charge Transfer Complexes for Organic Metals;Donors (Charge Transfer Complexes) | | Mol File: | 31366-25-3.mol | 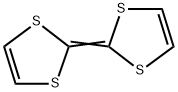 |
| | TETRATHIAFULVALENE Chemical Properties |
| Melting point | 116-119 °C (lit.) | | Boiling point | 90℃ (1 Torr) | | density | 1.448 (estimate) | | refractive index | 1.6000 (estimate) | | storage temp. | 2-8°C | | form | Crystals or Crystalline Powder | | color | Orange to brownish | | Water Solubility | It is insoluble in water. Soluble in organic solvents. | | Sensitive | Air & Light Sensitive | | λmax | 369nm(CHCl3)(lit.) | | Merck | 14,9242 | | BRN | 1617956 | | InChIKey | FHCPAXDKURNIOZ-UHFFFAOYSA-N | | CAS DataBase Reference | 31366-25-3(CAS DataBase Reference) | | EPA Substance Registry System | 1,3-Dithiole, 2-(1,3-dithiol-2-ylidene)- (31366-25-3) |
| | TETRATHIAFULVALENE Usage And Synthesis |
| Chemical Properties | ORANGE TO BROWNISH CRYSTALS OR CRYSTALLINE POWDER | | Uses | Electron donor for supramolecular synthesis,1 charge-transfer complex synthesis2 with 7,7,8,8-tetracyanoquinodimethane (cat. no. 157635), and for electron transfer to diazonium salts.3 | | Uses | Tetrathiafulvalene, holds wide application in HPLC, NMR. This heterocyclic compound contributed to the development of molecular electronics. They are used as a organic super conductors. | | Uses | Molecular sensors; radical catalyst. | | Definition | ChEBI: Tetrathiafulvalene is a member of the class of fulvalenes that is ethene substituted by 1,3-dithiol-2-ylidene groups at positions 1 and 2. It is an organosulfur heterocyclic compound and a member of fulvalenes. | | General Description | Tetrathiafulvalene (TTF) is an electron-donor which consists of oligomers, dendrimers and polymers which can be used in the formation of redox macromolecules. | | Purification Methods | Recrystallise it from cyclohexane/hexane under an argon atmosphere [Kauzlarich et al. J Am Chem Soc 109 4561 1987]. [Beilstein 19/11 V 380.] |
| | TETRATHIAFULVALENE Preparation Products And Raw materials |
|