|
|
| | TERT-BUTYL 4-BROMOBENZYLCARBAMATE Basic information |
| Product Name: | TERT-BUTYL 4-BROMOBENZYLCARBAMATE | | Synonyms: | TERT-BUTYL 4-BROMOBENZYLCARBAMATE;Carbamic acid, (4-bromophenyl)methyl-, 1,1-dimethylethyl ester;(4-BROMO-BENZYL)-CARBAMIC ACID TERT-BUTYL ESTER;4-Bromo-N-BOC-benzylamine;N-(4-Bromobenzyl)carbamic acid tert-butyl ester;tert-Butyl N-(4-bromobenzyl)carbamate;Carbamic acid, N-[(4-bromophenyl)methyl]-, 1,1-dimethylethyl ester;tert-Butyl 4-bromobenzylcarbamate 98% | | CAS: | 68819-84-1 | | MF: | C12H16BrNO2 | | MW: | 286.16 | | EINECS: | | | Product Categories: | amine|alkyl bromide| carboxylic ester | | Mol File: | 68819-84-1.mol | 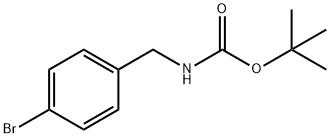 |
| | TERT-BUTYL 4-BROMOBENZYLCARBAMATE Chemical Properties |
| Melting point | 86-88℃ | | Boiling point | 374.3±25.0 °C(Predicted) | | density | 1.319±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | form | solid | | pka | 12.05±0.46(Predicted) | | color | Off-white |
| Hazard Codes | Xn | | Risk Statements | 22 | | HS Code | 2924297099 |
| | TERT-BUTYL 4-BROMOBENZYLCARBAMATE Usage And Synthesis |
| Synthesis | Tert-Butyl 4-bromobenzylcarbamate is prepared by the reaction of di-tert-butyl dicarbonate and 4-bromobenzylamine hydrochloride. The specific synthesis steps are as follows:
To a stirred suspension of 4-bromo-benzylamine hydrochloride (968mg, 4.35mmol) and triethylamine (0.666ml, 4.79mmol) in chloroform (15ml) was added di-tert-butyldicarbonate (949mg, 4.35mmol). The resulting solution was stirred at ambient temperature for 2h and then diluted with DCM (40ml). The organic solution was washed sequentially with 10% aqueous citric acid (40ml) and brine (40ml). The organic phase was dried over anhydrous magnesium sulfate and the filtrate evaporated at reduced pressure to afford Tert-Butyl 4-bromobenzylcarbamate (1.23g, 99%). 1H NMR 7.48-7.43 (2H, m), 7.18 (2H, d, 8.4), 4.84 (1H, br s), 4.27 (2H, d, 5.7), 1.46 (9H, s).
 |
| | TERT-BUTYL 4-BROMOBENZYLCARBAMATE Preparation Products And Raw materials |
|