|
|
| | 2-Bromo-3′-hydroxyacetophenone Basic information |
| Product Name: | 2-Bromo-3′-hydroxyacetophenone | | Synonyms: | 2-Bromo-3'-hydroxyacetophenone, 2-Bromo-1-(3-hydroxyphenyl)ethan-1-one, 3-(Bromoacetyl)phenol;M-BroMoacetylphenol;omega-Bromo-m-hydroxyacetophenone;Ethanone,2-broMo-1-(3-hydroxyphenyl)-;2-bromo-1-(3-hydroxyphenyl)ethan-1-one;Phenylephrine Impurity 64;Carteolol Impurity 13;2-Bromo-3-Hydroxyacetophenone (2-Bromo-3-HAP) | | CAS: | 2491-37-4 | | MF: | C8H7BrO2 | | MW: | 215.04 | | EINECS: | | | Product Categories: | Aromatics;Carbonyl Compounds;Ketones;C7 to C8 | | Mol File: | 2491-37-4.mol | 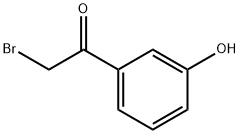 |
| | 2-Bromo-3′-hydroxyacetophenone Chemical Properties |
| Melting point | 70-74 °C(lit.) | | Boiling point | 327.8±22.0 °C(Predicted) | | density | 1.622±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | soluble in Dichloromethane, Ethyl Acetate, Methanol | | form | powder | | pka | 8.97±0.10(Predicted) | | color | Fawn to brown |
| Hazard Codes | C | | Risk Statements | 22-34-43 | | Safety Statements | 26-36/37/39-45 | | RIDADR | UN 3263 8/PG 2 | | WGK Germany | 3 | | HS Code | 29147000 |
| | 2-Bromo-3′-hydroxyacetophenone Usage And Synthesis |
| Chemical Properties | White Solid | | Uses | Reactant involved in the synthesis of:
- Pyrrole-3-carboxylic acids
- N-aryl-N-thiazolyl compounds via the Hantzsch reaction
- Disubstituted oxadiazolylindole derivatives with antiinflammatory, analgesic and nitric oxide releasing activity
- Anilino-aryl-thiazoles with inhibitory activity toward valosin-containing proteins
- Tyrosine kinase erythropoietin inhibitors
- E. coli methionine aminopeptidase inhibitors
|
| | 2-Bromo-3′-hydroxyacetophenone Preparation Products And Raw materials |
|